  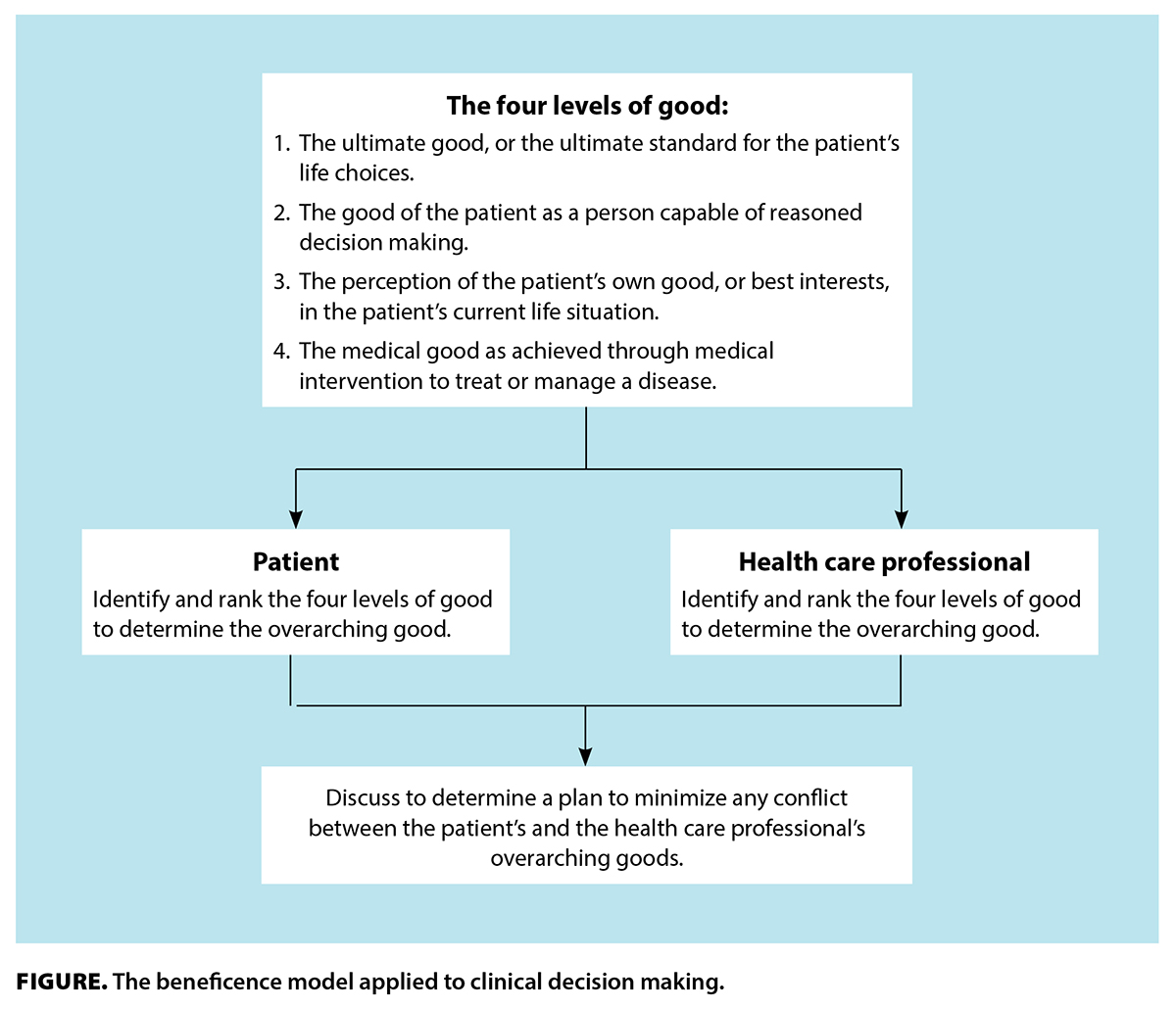
Further, during the risk/benefit analysis researchers need to keep in mind the impact of their work not just on participants, but also on nonparticipants and social systems. As the result of a risk/benefit analysis, a researcher could adjust the study design to reduce the probability of an adverse event (e.g., screen out participants who are vulnerable) or reduce the severity of an adverse event if it occurs (e.g., make counseling available to participants who request it). Analysis of risk should include two elements: the probability of adverse events and the severity of those events. This first process is largely a technical matter requiring substantive expertise, while the second is largely an ethical matter where substantive expertise may be less valuable, or even detrimental.Ī risk/benefit analysis involves both understanding and improving the risks and benefits of a study. In practice, the principle of Beneficence has been interpreted to mean that researchers should undertake two separate processes: a risk/benefit analysis and then a decision about whether the risks and benefits strike an appropriate ethical balance. Therefore, the imperative of not doing harm can be in conflict with the imperative to learn, leading researchers to occasionally make difficult decisions about “when it is justifiable to seek certain benefits despite the risks involved, and when the benefits should be foregone because of the risks” (Belmont Report 1979). However, the Belmont Report also acknowledges that learning what is beneficial may involve exposing some people to risk. The Belmont Report traces the idea of “do not harm” to the Hippocratic tradition in medical ethics, and it can be expressed in a strong form where researchers “should not injure one person regardless of the benefits that might come to others” (Belmont Report 1979). The Belmont Report argues that the principle of Beneficence is an obligation that researchers have to participants, and that it involves two parts: (1) do not harm and (2) maximize possible benefits and minimize possible harms. 7.2.2 Participant-centered data collectionīeneficence is about understanding and improving the risk/benefit profile of your study, and then deciding if it strikes the right balance.7.2.1 The blending of readymades and custommades.6.7.3 Think of research ethics as continuous, not discrete.6.7.2 Put yourself in everyone else’s shoes.6.7.1 The IRB is a floor, not a ceiling.6.6.4 Making decisions in the face of uncertainty. 
6.6.2 Understanding and managing informational risk.6.4.4 Respect for Law and Public Interest.5.2.2 Crowd-coding of political manifestos.4.6.2 Build ethics into your design: replace, refine, and reduce.4.4.2 Heterogeneity of treatment effects.4.3 Two dimensions of experiments: lab-field and analog-digital.2.3 Ten common characteristics of big data 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
